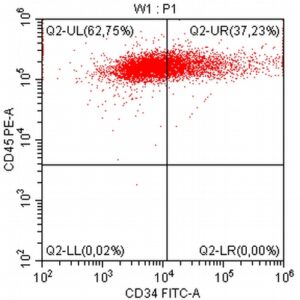
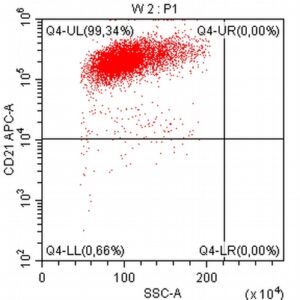
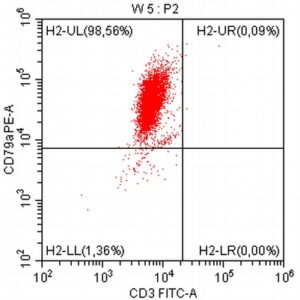
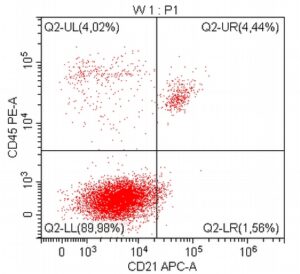
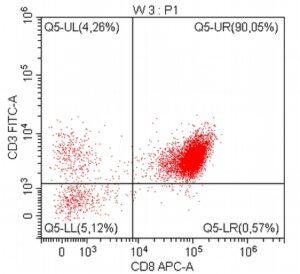
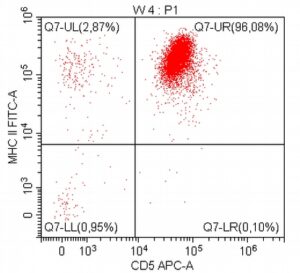
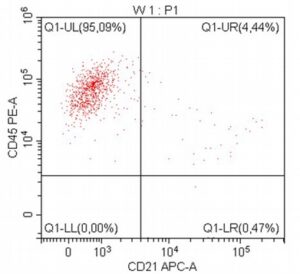
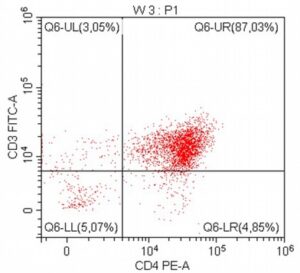
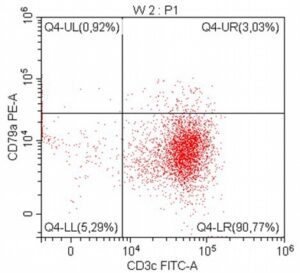
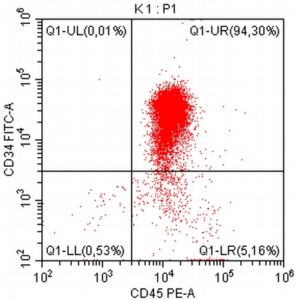
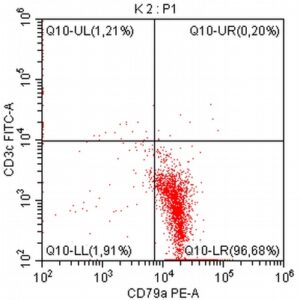
- A 10-year-old Schnauzer with clinical suspicion of lymphoma confirmed by cytopathological examination of the lymph nodes.
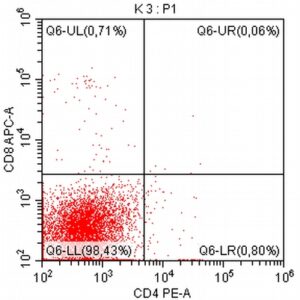
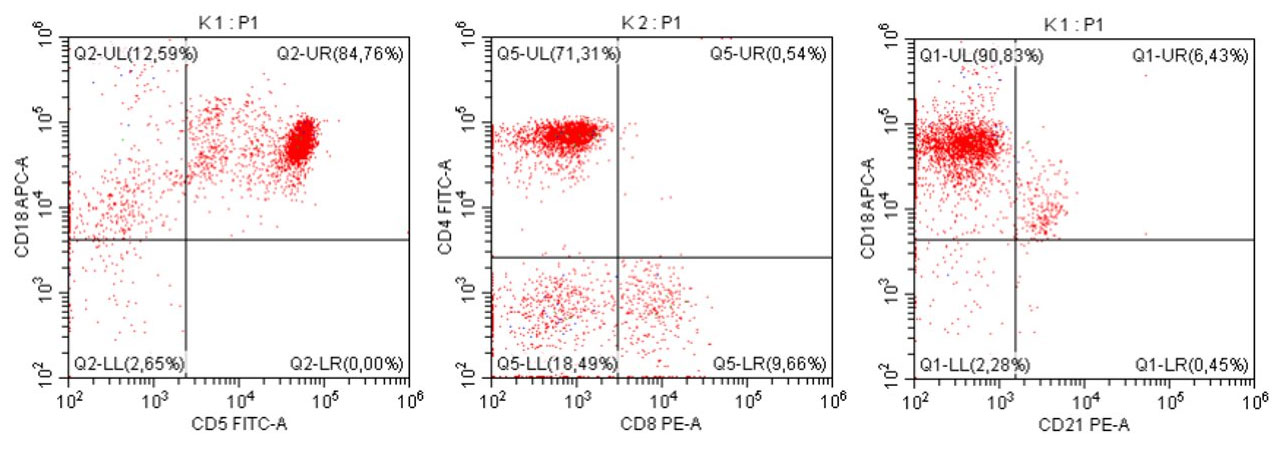
- To assess the clonality and origin of the neoplastic cells, a PARR test was performed, which surprisingly yielded a negative result (features of polyclonal proliferation of T and B lymphocytes were found, as seen in non-neoplastic stimulation of lymph nodes).
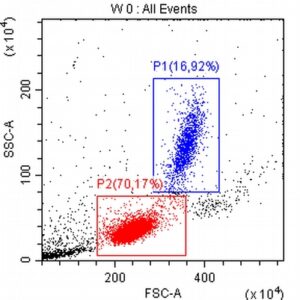
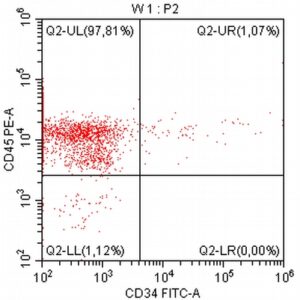
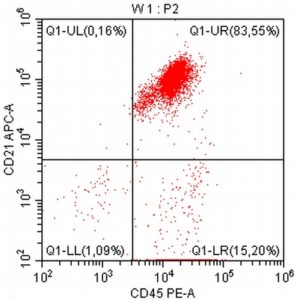
- The results of the cytometric analysis showed that the dominant population in the examined lymph node consisted of medium to large cells with the immunophenotype CD45+ CD21+ CD79α+ MHC II+. This finding indicated a disruption of the physiological proportions between T and B lymphocytes in the lymph node and clearly suggested excessive proliferation of B lymphocytes.*
Taking into account the clinical data and the cytology results of the patient’s lymph nodes, immunophenotyping made it possible to resolve doubts about the final diagnosis — the presence of B-cell lymphoma was confirmed and appropriate therapy was initiated.
*Pawlak A, et al. Immunophenotypic characterization of canine malignant lymphoma: a retrospective study of cases diagnosed in Poland Lower Silesia, over the period 2011–2013. Veterinary and Comparative Oncology, 2014, 14, 52–59.